
Contact Information
Urbana, IL 61801
Research Interests
Research Topics
Host-Pathogen Interactions, Imaging, Virology
Disease Research Interests
Infectious Diseases
Research Description
Mechanisms of HIV pathogenesis in animal models and human patient samples; development and application of multiscale tissue imaging methods
HIV, the causative agent of AIDS, was discovered over 35 years ago and remains a global health concern with an estimated 37 million individuals currently infected worldwide. Despite extensive efforts, there is currently no cure for HIV and although antiretroviral therapy allows individuals with access to medicine to lead normal lives, the presence of a reservoir of latently infected cells that rebound upon cessation of treatment requires infected individuals to remain on treatment indefinitely. My lab focuses on using advanced imaging techniques to visualize HIV pathogenesis in tissues with the goals of understanding relevant modes of virus dissemination, investigating the latent virus reservoir, and characterizing the effectiveness of anti-HIV therapies.
Relatively little is known about the mechanisms of HIV dissemination and the organization of the latent reservoir of HIV infected cells in human patients at the resolution of individual infected cells and virions within intact tissues. In order to address these limitations, my lab is utilizing a multiscale imaging approach to visualize mechanisms of HIV pathogenesis in tissues from small animal models of HIV infection at multiple levels of volume and resolution.
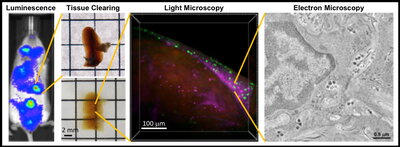
Mice with humanized immune systems are a tractable model for studying HIV infection in the presence of an immune response and recapitulate many aspects of disease in humans. In this model, human immune cells are introduced into immunodeficient mice and a “human-like” immune system is rapidly reconstituted within weeks to months. These “humanized” mice can be infected with HIV and be monitored longitudinally in order to understand the interactions between the virus and the immune system during specific stages of infection. For example, bioluminescence imaging allows whole animal imaging and can identify regions of active HIV replication within a living animal for further analysis with higher resolution methods.
For larger volume imaging with single-cell resolution we use tissue clearing and light microscopy. Tissue clearing involves treating tissues with chemical cocktails that remove opaque biomolecules that limit light penetration into the sample. These cleared tissues become optically transparent and can be labeled with fluorescent probes specific for actively infected cells, latently infected cells, and individual immune cell populations. Light microscopy of labeled tissues reveals the spatial distribution of individual HIV-infected cells and target cells within volumes (mm3-cm3) of intact tissue containing tens of thousands to millions of cells. For smaller volume imaging with single-virus resolution we use electron microscopy which generates higher resolution 3D information to reveal ultrastructural details of HIV-infected cells and individual viruses within tissues.
Combining these multiscale imaging techniques provides insights into mechanisms of HIV pathogenesis in tissues at previously unattainable levels of volume and resolution, reveals differences between mechanisms of infection in tissues compared to cultured cells, and can identify virus reservoirs or tissue sanctuaries at specific times during HIV infection. Importantly, this multiscale imaging approach is directly translatable to non-human primates and human patient samples that we are pursuing through established collaborations.
Education
B.S. (Bacteriology; Genetics) University of Wisconsin-Madison
Ph.D. (Biochemistry) University of Utah
Post-Doctoral (Oncology) University of Utah
Post-Doctoral (Biology and Biological Engineering) Caltech
Additional Campus Affiliations
External Links
Highlighted Publications
Representative Publications
Zhang T, Gupta A, Frederick D, Layman L, Smith DM, Gianella S, Kieffer C. “3D Visualization of Immune Cell Populations in HIV-Infected Tissues via Clearing, Immunostaining, Confocal, and Light Sheet Fluorescence Microscopy.” Journal of Visualized Experiments. Published online May 6, 2021 doi: 10.3791/62441.
Hoffman MAG, Kieffer C, Bjorkman PJ. “In vitro Characterization of engineered red blood cells as potent viral traps against HIV-1 and SARS-CoV-2.” Molecular Therapy – Methods & Clinical Development. Published online Mar 10, 2021 doi:10.1016/j.omtm.2021.03.003
Ladinsky MS, Khamaikawin W, Jung Y. Lin S, Lam J, An DS, Bjorkman PJ, Kieffer C. Mechanisms of virus dissemination in bone marrow of HIV-1–infected humanized BLT mice. eLife 2019; 8:e46916.doi:10.7554/eLife.46916
Ventura, JD, Beloor, J, Allen, E, Zhang, T, Haugh, KA, Uchil, PD, Ochsenbauer, C, Kieffer, C, Kumar, P, Hope, TJ, Mothes, W. Longitudinal bioluminescent imaging of HIV-1 infection during antiretroviral therapy and treatment interruption in humanized mice. PLoS Pathog 2019; 15(12): e1008161.
Kieffer, C, Ladinsky MS, Bjorkman, PJ “Multiscale imaging of HIV-1 transmission in humanized mice. AIDS Res Hum Retroviruses, Published online Sep 7, 2017. doi:10.1089/AID.2017.0142
*Cover article
Kieffer C, Ladinsky MS, Ninh A, Galimidi RP, Bjorkman PJ "Longitudinal imaging of HIV-1 spread in humanized mice with parallel 3D immunofluorescence and electron tomography." eLIFE, published online February 15, 2017, DOI: http://dx.doi.org/10.7554/eLife.23282.
Ladinsky, MS, Kieffer C, Olson, G, Deruaz, M, Vrbanac, V, Tager, AM, Kwon, DS, Bjorkman, PJ "Electron Tomography of HIV-1 Infection in Gut-Associated Lymphoid Tissue" PLoS Pathog 2014, Jan 10(1): e1003899. DOI: 10.1371/journal.ppat.1003899
*Basham, KJ, *Kieffer, C, Shelton, DN, Leonard, CJ, Bhonde, VR, Vankayalapati, H, Milash, B, Bearss, DJ, Looper, RE, Welm, BE "Chemical Genetic Screen Reveals a Role for Desmosomal Adhesion in Mammary Branching Morphogenesis." J Biol Chem 2013 Jan 25; 288(4):2261-70.
* Authors contributed equally
Kieffer, C, Skalicky, JJ, Morita, E, De Domenico, I, Ward, DM, Kaplan, J, Sundquist, WI. “Two distinct modes of ESCRT-III recognition are required for VPS4 functions in lysosomal protein targeting and HIV-1 budding.” Developmental Cell, 2008 Jul; 15(1):62-73.
Stuchell-Brereton, MD, Skalicky JJ, Kieffer, C, Karren, MA, Ghaffarian S, Sundquist, WI. “ESCRT-III recognition by VPS4 ATPases.” Nature, 2007 Oct 11;449(7163):740-4.
Recent Publications
Smith, D. M., Rowland, E. F., Gianella, S., Patel, S. P., Solso, S., Dullano, C., Deiss, R., Wells, D., Ignacio, C., Caballero, G., Porrachia, M., Kieffer, C., & Chaillon, A. (2025). HIV Integration into the PTEN Gene and Its Tumor Microenvironment Implications for Lung Cancer. Current Oncology, 32(7), Article 389. https://doi.org/10.3390/curroncol32070389
Blanco, A., Coronado, R. A., Arun, N., Ma, K., Dar, R. D., & Kieffer, C. (2024). Monocyte to macrophage differentiation and changes in cellular redox homeostasis promote cell type-specific HIV latency reactivation. Proceedings of the National Academy of Sciences of the United States of America, 121(19), Article e2313823121. https://doi.org/10.1073/pnas.2313823121
Rivera-Cardona, J., Kakuturu, N., Rowland, E. F., Teo, Q. W., Thayer, E. A., Tan, T. J. C., Sun, J., Kieffer, C., Wu, N. C. H., & Brooke, C. B. (2024). Seasonal influenza a virus lineages exhibit divergent abilities to antagonize interferon induction and signaling. PLoS pathogens, 20(12), Article e1012727. https://doi.org/10.1371/journal.ppat.1012727
Tan, T. J. C., Mou, Z., Lei, R., Ouyang, W. O., Yuan, M., Song, G., Andrabi, R., Wilson, I. A., Kieffer, C., Dai, X., Matreyek, K. A., & Wu, N. C. (2023). High-throughput identification of prefusion-stabilizing mutations in SARS-CoV-2 spike. Nature communications, 14(1), Article 2003. https://doi.org/10.1038/s41467-023-37786-1
Ouyang, W. O., Tan, T. J. C., Lei, R., Song, G., Kieffer, C., Andrabi, R., Matreyek, K. A., & Wu, N. C. (2022). Probing the biophysical constraints of SARS-CoV-2 spike N-terminal domain using deep mutational scanning. Science Advances, 8(47), Article eadd7221. https://doi.org/10.1126/sciadv.add7221




