
In a recently published study from the University of Illinois, researchers have identified a new class of bacterial importers in over 250 species.
Much like humans eat food in order to obtain essential nutrients, bacteria acquire nutrients by importing them. An essential nutrient for life is zinc, which cannot be manufactured, and therefore must be obtained from the environment. However, the availability of zinc is frequently limited. This is exemplified by a defense mechanism, nutritional immunity, used by the immune system to prevent infections in which the body withholds metals, such as zinc, to combat invading bacteria.
One recently discovered strategy that bacteria use to overcome nutritional immunity is the production of small zinc-binding molecules known as zincophores. These molecules which bind zinc tightly are produced by the bacterium, exported empty from the cell, and then imported back into the cell bound to zinc. Previous research revealed that the human pathogen Staphylococcus aureus and a handful of other bacteria use zincophores to obtain zinc, yet the full range of microbes that utilize these metal-binding molecules remained unknown.
A new study by postdoctoral researcher Jacqueline Morey and Thomas Kehl-Fie, a professor of microbiology, reveals the astounding prevalence and biodiversity of zincophores. Their article, “Bioinformatic Mapping of Opine-Like Zincophore Biosynthesis in Bacteria,” was published in mSystems. Using bioinformatics and computational approaches, they leveraged information about the machinery that produces zincophores in S. aureus and three other species of zincophore-producing bacteria to identify 250 additional species capable of producing these zinc acquisition molecules. Their work also revealed a previously unappreciated potential chemical diversity.
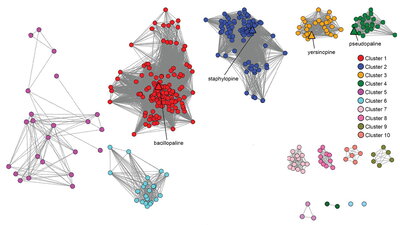
Researchers began their search for zincophore biosynthetic gene clusters (BGCs), the instructions for building the zincophore synthesis machinery, by searching databases for proteins that look similar to the zincophore synthesis machinery in S. aureus. Because proteins may look similar but participate in different processes, researchers next examined the surrounding genes to see if the produced proteins involved in zincophore synthesis, secretion, or import. They then examined the collection of genes in these loci identifying BGCs, in the process identifying clusters that contain extra enzymes. This suggested that the bacteria produce a wide range of similar but distinct zincophores.
The resulting list of bacterial species capable of producing zincophores includes microorganisms from the soil, sea, and plants as well as human and animal pathogens. Amongst the human and animal pathogens are bacteria that frequently cause infections in individuals with cystic fibrosis, wounds, and burns. This study not only sheds light on the biodiversity of zincophores among bacteria, but it also opens doors for researchers interested in investigating these molecules for their potential role in therapeutics to treat infection or tools for manipulating the soil microbiome to increase crop yield.