
Contact Information
University of Illinois at Urbana-Champaign
C422 Chemical and Life Sciences Laboratory
601 S. Goodwin Avenue
Urbana, IL 61801
Research Interests
Research Topics
Chromatin Structure, DNA Biology, Protein-Nucleic Acid Interactions
Disease Research Interests
Cancer
Research Description
Eukaryotic DNA replication; Chromosome structure & maintenance; Heterochromatin organization; Cell cycle control
The initiation of DNA replication in eukaryotic cells is a highly regulated process that leads to the duplication of genetic information for the next cell generation. DNA replication, which occurs during S phase of the cell cycle, is intimately linked to mitotic progression and eventually cell division. Inaccurate DNA replication in turns leads to abnormal chromosome segregation resulting in aneuploidy and genomic instability, a hallmark of most cancerous cells. Thus the accurate duplication of DNA is of paramount importance and is governed by a number of proteins including the Origin Recognition complex (ORC) which serves as a landing pad for the assembly of a multiprotein pre-replicative complex. Other than its bonafide role in DNA replication, ORC proteins are involved in diverse functions including gene silencing, heterochromatin organization, cytokinesis and also in dendrite formation in postmitotic neurons. The focal point of research in my lab is to study the events and uncover the cues that integrate DNA replication with heterochromatin organization, chromosome segregation and cytokinesis, major focus being on the role of ORC in interconnecting these events.
The research project includes:
Role of ORC proteins in heterochromatin organization and chromosome structure
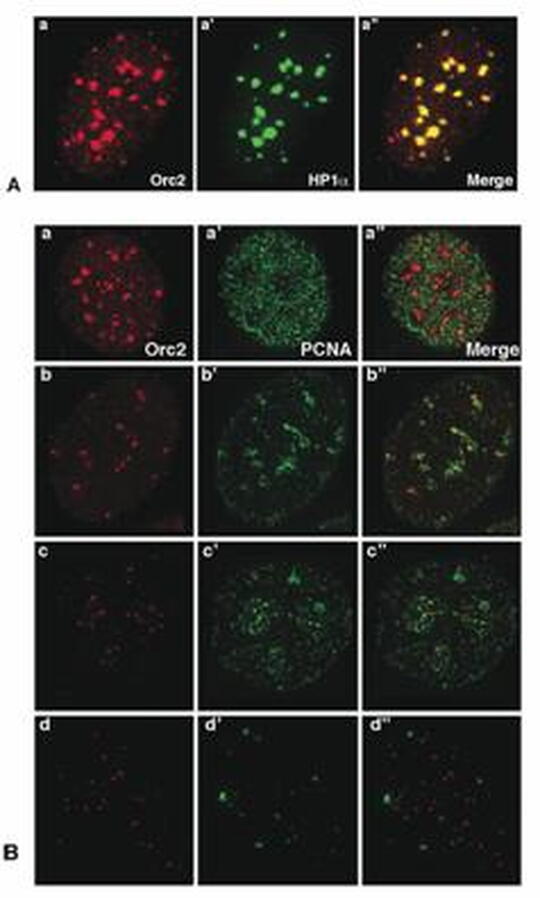
A. Colocalization of Orc2 and HP1 in MCF7 cells at the heterochromatin
B. Differential binding of Orc2 to the heterochromatin.
Prasanth et al., 2004 EMBO J. 23(13)2651-6
ORC proteins have been implicated in heterochromatin organization in mammalian cells (Figure 1) and Drosophila and gene silencing in Saccharomyces cerevisiae. To further pinpoint the role of ORC subunits at heterochromatin, my lab focuses on the identification of the interacting partners of ORCs at heterochromatin during different stages of the cell cycle and to delineate the replication function of ORC proteins versus their role in chromatin compaction.
It has been suggested that centromeric heterochromatin is necessary for both the cohesion of sister chromatids and the subsequent normal disjunction of mitotic chromosomes. In the fission yeast Schizosaccharomyces pombe, the homologue of the heterochromatin protein (HP1) protein Swi6 is critical for efficient sister chromatid cohesion during cell division. The loss of Orc2 and Orc3 proteins from human cells also results in the loss of HP1 (HP1a and b) from the centric heterochromatin during mitosis. In Orc2 depleted cells, a population of cells arrests in mitosis with defective chromosome structure and condensation, spindle and chromosome congression defects.
How the Orc2 depletion results in the abnormal localization of HP1 proteins from mitotic chromosomes remains to be addressed. Studying the localization of proteins which interact with ORC at heterochromatin with respect to differential levels of ORC proteins as well as dissecting the functional significance of their interaction will be critical towards understanding how ORC is involved in organizing chromatin structure during different stages of the cell-cycle. Further, live cell imaging using mitotic markers will enable to study the order of the mitotic defects in ORC depleted cells.
Also, structure function analysis of ORC and the heterochromatin interacting partners would provide better insights into the understanding of ORCs involvement in heterochromatin organization and chromosome structure.
Role of Orc6 proteins in cell-cycle progression and cytokinesis
The second main area of the research focus of my lab is to define the role of the replication initiator protein Orc6 in cytokinesis and its interaction with proteins that govern the chromosome and cell division cycle in human cells.
Although Orc6 depletion results in abnormal cytokinesis, how Orc6 functions in coordinating chromosome segregation and cytokinesis is not understood. Proteins interacting with Orc6 during different stages of the cell-cycle would throw light on how Orc6 is involved in cell-cycle progression. Further, the mechanism that results in cytokinesis defects in the absence of Orc6 and the stages at which Orc6 executes its function remains to be elucidated. Time-lapse imaging experiments will be used to investigate the mechanism of Orc6 function. Further, genetic complementation experiments will be used to delineate the replication function and the cytokinesis function of Orc6 in human cells.
ORC and other human replication initiation proteins have been shown to be required for replication of Epstein Barr virus (EBV), a virus that is associated with Burkitt’s lymphoma, immunoblastic B-cell lymphoma and Hodgkin’s diseas. Deletion of ORC genes has been observed in non-Hodgkin’s lymphomas, uterine leiomyomas and malignant myeloid disease. By increasing our understanding of the involvement of ORC in critical cellular processes including cell chromosome duplication and segregation, we will gain valuable insight into how genetic instability contributes to cancer progression.
Education
M.Sc., University of Delhi (Genetics), New Delhi, India
Ph.D., National Institute of Immunology (Molecular Genetics and Life Sciences), New Delhi, India
Postdoctoral Fellow (Special Fellow, Leukemia and Lymphoma Society), Cold Spring Harbor Laboratory-New York, USA
Awards and Honors
National Science Foundation Career Award
Lynn M. Martin Award for distinguished women teachers
MCB Research Excellence Award
Campus Distinguished promotion award
University Scholar
Courses Taught
Additional Campus Affiliations
External Links
Highlighted Publications
Rosaline Y.C. Hsu, Yo-Chuen Lin, Christophe Redon, Qinyu Sun, Deepak K. Singh, Yating Wang, Vasudha Aggarwal, Jaba Mitra, Abhijith Matur, Branden Moriarity, Taekjip Ha, Mirit I Aladjem, Kannanganattu V. Prasanth and Supriya G. Prasanth (2020) ORCA/LRWD1 regulates homologous recombination at ALT-telomeres by modulating heterochromatin organization. iScience, April 17: https://doi.org/10.1016/j.isci.2020.101038
Yo-Chuen Lin+, Yating Wang+, Rosaline Hsu, Sumanprava Giri, Susan Wopat, Mariam K. Arif, Arindam Chakraborty, Kannanganattu V. Prasanth, and Supriya G. Prasanth*: PCNA-mediated stabilization of E3 ligase RFWD3 at the replication fork is essential for DNA replication. PNAS (USA) 2018 Dec 10. pii: 201814521. doi: 10.1073/pnas.1814521115. [Epub ahead of print]
Wang Y, Khan A, Marks AB, Smith OK, Giri S, Lin YC, Creager R, MacAlpine DM, Prasanth KV, Aladjem MI, Prasanth SG. (2017). Tempral association of ORCA/LRWD1 to late-firing origins during G1 dictates heterochromatin replication and organization. Nucleic Acids Res, pii: gkw1211. PMCID: PMC5389698
Sumanprava Giri, Arindam Chakraborty, Kizhakke M. Sathyan K, Kannanganattu V. Prasanth and Supriya G. Prasanth (2016).Orc5 induces large-scale chromatin decondensation in a GCN5-dependent manner. J. Cell Sci., Jan 15;129(2):417-29.
Abid Khan, Sumanprava Giri, Yating Wang, Arindam Chakraborty, Archit K Ghosh, Aparna Anantharaman, Vasudha Aggarwal, Kizhakke M Sathyan, Taekjip Ha, Kannanganattu V Prasanth and Supriya G Prasanth (2015). BEND3 represses rDNA transcription by stabilizing a NoRC component via USP21 deubiquitinase. PNAS(USA), Jul 7;112(27):8338-43. doi: 10.1073/pnas.1424705112.
Sumanprava Giri, Vasudha Aggarwal, Julien Pontis, Zhen Shen, Arindam Chakraborty, Abid Khan, Craig Mizzen, Kannanganattu V. Prasanth, Slimane Ait-Si-Ali, Taekjip Ha and Supriya G. Prasanth (2015). The preRC protein ORCA organizes heterochromatin by assembling histone H3 lysine 9 methyltransferases on chromatin. eLIFE Apr 29. 10.7554/eLife.06496
Arindam Chakraborty, Kannanganattu V. Prasanth and Supriya G. Prasanth (2014). Dynamic phosphorylation of HP1a regulates mitotic progression in human cells. Nature Communications. DOI: 10.1038/ncomms4445.
Shen Z, Chakraborty A, Jain A, Giri S, Ha T, Prasanth KV and Supriya G. Prasanth (2012). Dynamic association of ORCA with preRC components regulates DNA replication initiation. Mol Cell Biol. 32(15): 3107-3120.
Zhen Shen, Kizhakke M. Sathyan, Yijie Geng, Ruiping Zheng, Arindam Chakraborty, Brian Freeman, Fei Wang, Kannanganattu V. Prasanth and Supriya G. Prasanth (2010) A novel WD-repeat protein stabilizes ORC binding to chromatin. Molecular Cell2010 Oct. 4; 40(1): 99-111 (PMID: 20932478).
Supriya G. Prasanth*, Zhen Shen, Kannanganattu V. Prasanth and Bruce Stillman* (2010). Human ORC is essential for HP1 binding to chromatin and for heterochromatin organization. PNAS, Aug 24;107(34):15093-8. Epub 2010 Aug 5. (* first and co-corresponding author) [FULL TEXT PDF]
Adriana Hemerly, Supriya G. Prasanth, Khalid Siddiqui and Bruce Stillman (2009). Orc1 Controls Centriole and Centrosome Copy Number in Human Cells. Science, Feb 6, 323: 789-793. [FULL TEXT PDF]
Supriya G. Prasanth, Kannanganattu V. Prasanth, Khalid Siddiqui, David L. Spector and Bruce Stillman (2004). Human Orc2 localizes to centrosomes, centromeres and heterochromatin during chromosome inheritance. EMBO J. Jul 7;23(13):2651-6.
Supriya Gangadharan Prasanth, Kannanganattu V. Prasanth and Bruce Stillman (2002). Orc6 involved in DNA replication, chromosome segregation and cytokinesis. Science 297(5583): 1026-1031.
Recent Publications
Song, Y. J., Shinn, M. K., Bangru, S., Wang, Y., Sun, Q., Hao, Q., Chaturvedi, P., Freier, S. M., Perez-Pinera, P., Nelson, E. R., Belmont, A. S., Guttman, M., Prasanth, S. G., Kalsotra, A., Pappu, R. V., & Prasanth, K. V. (2026). LncRNA-splicing factor condensates regulate hypoxia-responsive pre-mRNA processing near nuclear speckles. Molecular cell, 86(6), 1061-1080.e10. https://doi.org/10.1016/j.molcel.2026.02.014
Ahmad, H., Chetlangia, N., & Prasanth, S. G. (2024). Chromatin’s Influence on Pre-Replication Complex Assembly and Function. Biology, 13(3), Article 152. https://doi.org/10.3390/biology13030152
Hao, Q., Liu, M., Daulatabad, S. V., Gaffari, S., Song, Y. J., Srivastava, R., Bhaskar, S., Moitra, A., Mangan, H., Tseng, E., Gilmore, R. B., Frier, S. M., Chen, X., Wang, C., Huang, S., Chamberlain, S., Jin, H., Korlach, J., McStay, B., ... Prasanth, K. V. (2024). Monoallelically expressed noncoding RNAs form nucleolar territories on NOR-containing chromosomes and regulate rRNA expression. eLife, 13, Article e80684. https://doi.org/10.7554/eLife.80684
Kurniawan, F., Chakraborty, A., Oishi, H. Z., Liu, M., Arif, M. K., Chen, D., Prasanth, R., Lin, Y. C., Olalaye, G., Prasanth, K. V., & Prasanth, S. G. (2024). Phosphorylation of Orc6 During Mitosis Regulates DNA Replication and Ribosome Biogenesis. Molecular and cellular biology, 44(7), 289-301. https://doi.org/10.1080/10985549.2024.2356880
Singh, D. K., Cong, Z., Song, Y. J., Liu, M., Chaudhary, R., Liu, D., Wang, Y., Prasanth, R., K C, R., Lizarazo, S., Akhnoukh, M., Gholamalamdari, O., Moitra, A., Jenkins, L. M., Bhargava, R., Nelson, E. R., Van Bortle, K., Prasanth, S. G., & Prasanth, K. V. (2024). MANCR lncRNA Modulates Cell-Cycle Progression and Metastasis by Cis-Regulation of Nuclear Rho-GEF. Molecular and cellular biology, 44(9), 372-390. https://doi.org/10.1080/10985549.2024.2383773




